Gas Chromatograph Contamination Issues
In the last article, the basics of gas chromatography- mass spectrometry (GC-MS) were explored. This article will address the importance of proper maintenance of the gas chromatograph. If improperly maintained, gas chromatographs become dirty with the residue from multiple drug tests. This residue can cause test contamination, which will likely affect the laboratory results in your client’s drug case.
In order to test drug samples, a chemist must take a sample of the seized drug and prepare it for the GC-MS testing. This prepared sample is placed into a vial. A portion of the sample is extracted by a needle and then injected into the injector port for the gas chromatograph. The sample is instantly vaporized by heat inside the injector port. Once the drug sample is vaporized inside the inlet, then the sample is carried into the gas chromatograph column.
The inlet has a liner that allows the smooth flow of the sample from the injection port into the column. (See Figure 1 below). Typical liners are made of glass with a chemical coating. (See Figure 2 below). Testing solid drugs is a “dirty” process that will eventually leave residue on the liner.
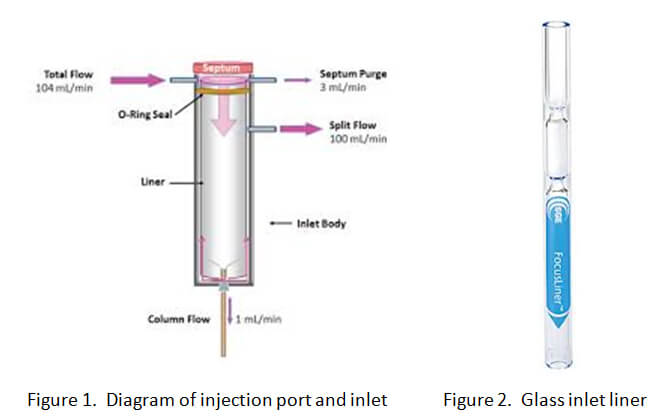
Dirty inlet liners will have a direct impact on drug analysis results. When numerous “dirty” samples have been analyzed, inlet liners must be replaced or cleaned routinely in order to minimize the influence of contaminants on subsequent drug sample analyses. Think of a dirty liner like a dirty barbeque grill. If you are heating chicken on the grill, residue from the chicken will remain until the grill is cleaned. This residue will be mixed with whatever food is next cooked on that dirty grill.  The chemical compounds contaminating a dirty liner can remain on the liner for extended periods of time and then elute during the testing of your client’s seized drug sample (urine or blood sample as well).
The chemical compounds contaminating a dirty liner can remain on the liner for extended periods of time and then elute during the testing of your client’s seized drug sample (urine or blood sample as well).
Another source of contamination can reside on the gold seal that is located inside the injection port. (See Figure 3). This gold seal can also collect residue from tests that can sit inside the seal and then later elute. The gold seal must be replaced or cleaned routinely.
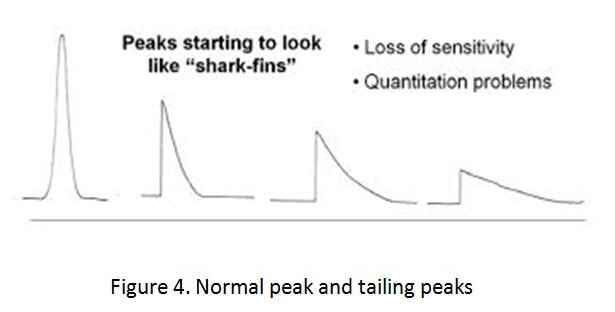
There are symptoms of a dirty liner that can be detected by thoroughly reviewing the analytical data. First, the chromatograms that are produced during the testing of the drug sample will show extraneous peaks. Second, the chromatographic peaks will have a tail, instead of being symmetrical. (See Figure 4 below).
Good laboratory practices should be in place to ensure that carry-over of contaminants from one sample to the next is eliminated. The gold seal and inlet liner should be cleaned or replaced on a weekly basis, if not sooner. More important, the analytical method established by the laboratory should require that a clean or blank sample is inserted into the GC-MS between every sample tested. This blank sample can either be air or deionized water.
If a proper blank was used between test samples and there was no contamination carry-over, then a chromatogram can be produced by the laboratory showing that no extraneous compounds appeared during the analysis of the blank. However, if a peak(s) shows up on the chromatogram for the blank that was tested, then there is proof that the gas chromatograph was contaminated by an extraneous, carry-over compound. Without running blanks between tests of drug samples, the laboratory is not utilizing accepted scientific practices.

